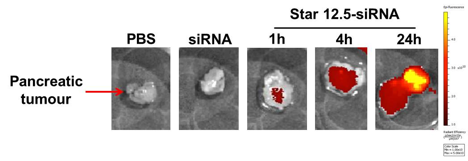
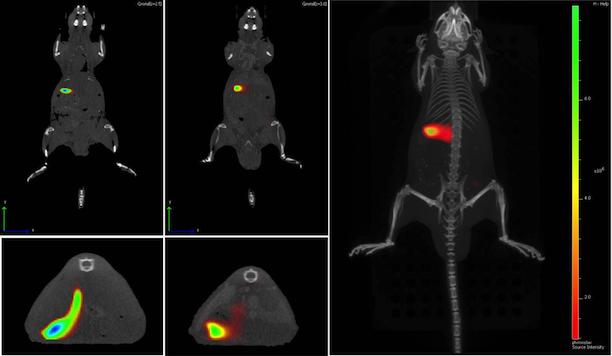
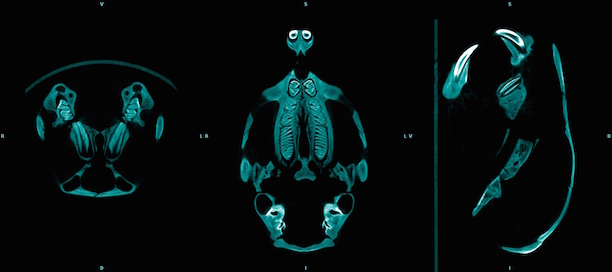
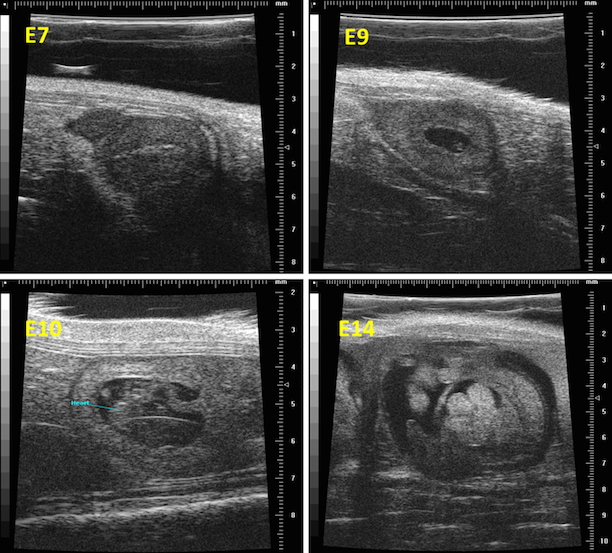
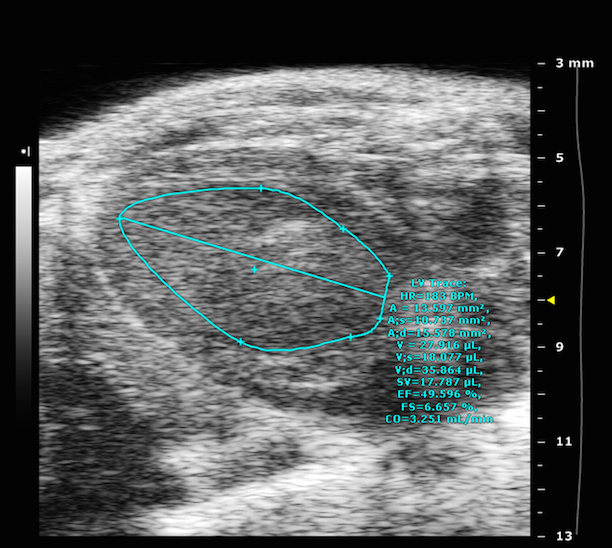
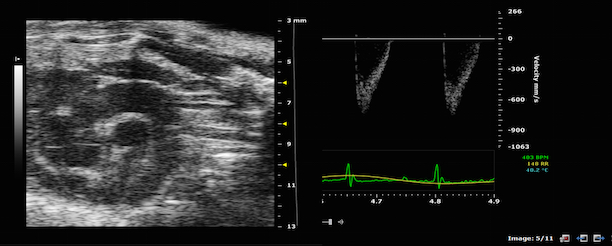
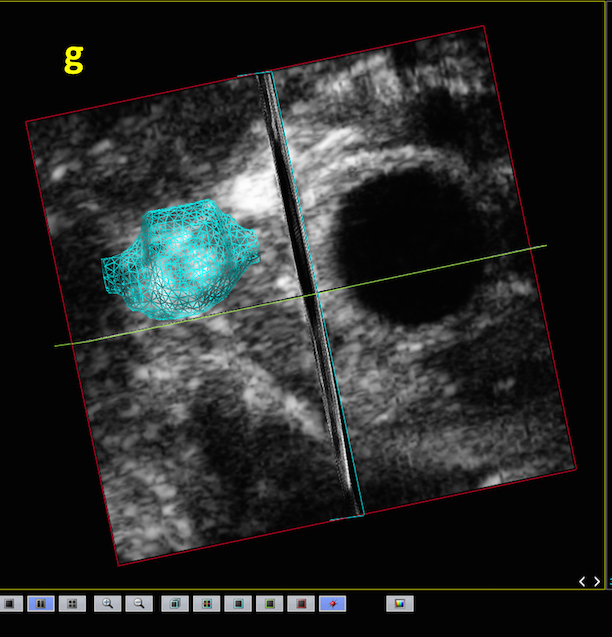
Magnetic Resonance Imaging (and Spectroscopy)
Magnetic Resonance Imaging (MRI) is based on the resonance response from magnetic atomic nuclei to radiofrequency excitation. Proton MRI utilizes 1H nuclei in water and other compounds to produce images that yield unique information about the local distribution and relaxation properties of water in tissues and samples. With its highly adaptable, proton-property dependent signal, MRI is able to produce exceptional image contrast in soft, moist tissues and biological samples.
In addition to its high-resolution structural imaging capabilities MRI is also capable to assess a wide variety of biomarkers that provide in-depth physiological and molecular information about tissue viability, function and therapy effects. MRI Methods for physiological and molecular imaging include, tissue vascularization imaging (e.g. perfusion, blood flow, vessel size), oxygenation mapping, localized metabolite assessment using spectroscopy, diffusion imaging, etc. Apart from protons, MRI can also use other nuclei to extend the spectrum of available physiological information. Examples are 3-He/129-Xe imaging, for lung ventilation studies, 31-P/13-C MRI/MRS for the assessment of metabolic pathways and 19-F imaging to assess local tissue oxygenation or cell tracking and targeted imaging.
UNSW BRIL runs a Bruker high-field pre-clinical MRI which is especially optimized for high-resolution imaging and assessment of physiological and molecular biomarkers in small samples and animals.
The Method – General Overview
Magnetic Resonance Imaging (MRI) is an imaging technology that is based on the resonance response from certain, spinning atomic nuclei to radiofrequency irradiation. Although a wide variety of atomic nuclei can be utilized to obtain information from experimental objects, in the vast majority of cases the response of protons (in water and certain metabolites) is used to produce images of tissues or samples. Proton MRI yields unique information about the local distribution of tissue/sample water and is able to deliver the best contrast for soft tissues of any imaging modality. Modern pre-clinical high field scanners are able to deliver high-resolution anatomical information that is useful for a wide range of applications [1].
Besides high-resolution, high-contrast anatomical imaging of soft-tissues, there is also a wide variety of MRI methods that are able to provide in-depth physiological information about the viability and function of tissues, organs and diseases.
Some examples of current physiological proton MRI methods and applications are:
· Diffusion of water in samples and tissue: Diffusion weighted MRI is able to give information about diffusion in living tissues. This can yield information about structure sizes in certain regions of interest as well as main anisotropy and directions of structures. This can be utilized to measure integrity of brain white matter axons, etc. [2]
· Tissue Vascularization: MRI can be used to gather a variety of information about the vascular system. Either contrast agent based or non-contrast agent based methods can be used to measure local blood supply, blood flow, blood volume or perfusion. Also vessel sizes and vessel permeability can be assessed with appropriate MRI methods.
· Flow imaging: MRI can be used to assess information about general flowing systems. Flow and flow velocities can be quantified over time [3].
· Fat imaging: Certain contrasts in MRI can be used to assess local fat distribution in tissue. Either contiguous fat pads can be assessed in size, or the local distribution of fat relative to water/muscle can be assessed on a pixel by pixel basis.
· Spectroscopy/CSI: MRI can be used to gather localized information about a wide variety of metabolites that contain protons with certain shifted resonances. Examples of metabolites that can be identified and quantified by proton MRI are NAA, Creatine, Choline, Glutamate, Glutamine, myo-inositol, etc. With so called spectroscopic imaging methods (also called chemical shift imaging, CSI) it is also possible to produce images of the distribution of single metabolite concentrations.
· Tissue oxygenation and oxygen supply can be assessed by exploiting specific magnetic properties of blood for MR Imaging. As blood iron changes its state from a paramagnetic to a diamagnetic state upon oxygenation information can be extracted about current oxygenation status of organs. This so-called BOLD (blood oxygenation level dependent) effect is used in neurofunctional MRI to map neuronal activity and can also deliver information about oxygen supply, e.g. in tumors [4].
· Magnetic properties of tissues and magnetization transfer can be exploited to collect information about local tissue integrity, e.g. in white matter myelin [5].
· Novel chemical exchange transfer saturation (CEST) imaging methods can be used to generate novel contrast agents to obtain information about further physiological parameters, such as local tissue pH [6].
Apart from protons, MRI can also use other nuclei that show nuclear resonances to extend the spectrum of available information. E.g. the following Isotopes can be used if special hardware is available (not generally the case for the UNSW facility): Phosphourous P, 3-He, 13-C, 129-Xe, 19F
These so called X-nuclei can open the field to interesting novel experiments and make additional information available, which is not otherwise accessible. Possible applications/examples of X-nuclei Imaging can be: Lung studies ventilation with noble gases as 3-He or 129-Xe, spectroscopy and imaging of metabolic pathways using 31-P od 13-C as “tracers”, oxygenation imaging, cell tracking and targeted imaging with 19-F nuclei, etc. Some examples may be found in the references [7], [8].
References
1. Benveniste, H. and S.J. Blackband, Translational neuroscience and magnetic-resonance microscopy. The Lancet Neurology, 2006. 5(6): p. 536-544.
2. Jiang, Y. and G.A. Johnson, Microscopic diffusion tensor imaging of the mouse brain. Neuroimage, 2010. 50(2): p. 465-471.
3. Lotz, J., et al., Cardiovascular flow measurement with Phase-Contrast MR imaging: Basic facts and implementation1. Radiographics, 2002. 22(3): p. 651-671.
4. He, X. and D.A. Yablonskiy, Quantitative BOLD: mapping of human cerebral deoxygenated blood volume and oxygen extraction fraction: default state. Magnetic Resonance in Medicine, 2007. 57(1): p. 115-126.
5. Liu, C., et al., High-field (9.4 T) MRI of brain dysmyelination by quantitative mapping of magnetic susceptibility. Neuroimage, 2011. 56(3): p. 930-938.
6. van Zijl, P. and N.N. Yadav, Chemical exchange saturation transfer (CEST): what is in a name and what isn't? Magnetic Resonance in Medicine, 2011. 65(4): p. 927-948.
7. Lilburn, D.M., G.E. Pavlovskaya, and T. Meersmann, Perspectives of Hyperpolarized Noble Gas MRI beyond< sup> 3</sup> He. Journal of Magnetic Resonance, 2012.
8. Ruiz‐Cabello, J., et al., Fluorine (19F) MRS and MRI in biomedicine. NMR in Biomedicine, 2011. 24(2): p. 114-129.